 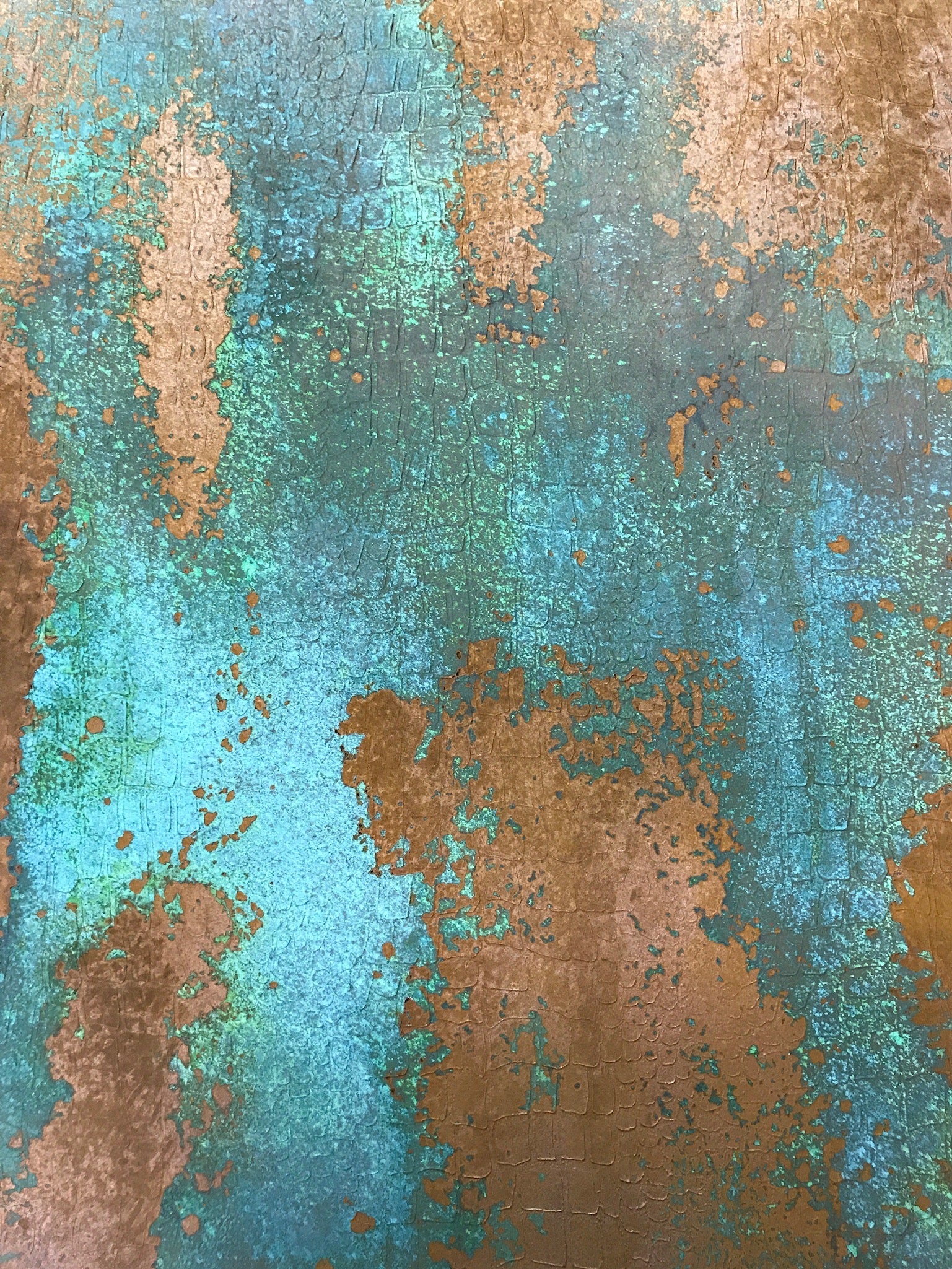
Rust starts to chip away and corrode the metal surface, creating porous cracks and hazardous, sharp edges. Rust is caused by deterioration, whereas tarnish and patina are made through oxidation.It’s also quite a bit more dangerous in most situations.īelow, you’ll find the five main reasons that make rust, tarnish, and patina so different from one another: Rust is the outlier of the group since it’s formed through a separate process and with different types of metal. 
Patina is simply a form of tarnish, which makes it the same thing in a sense. How Is Rust Different From Tarnish and Patina?Īs mentioned above, tarnish, and patina are very similar. How Is Rust Different From Tarnish and Patina?.Some examples of products with a patina finish include copper roofs, bronze statues, and brass door knockers. Some examples of products with a rust finish include iron fences, steel gutters, and cast iron pans. There are many products that are made with a rust or patina finish. Examples of products with a rust or patina finish This will cause the metal to react with the oxygen in the air to form a reddish-brown oxide coating on the surface of the metal. How to create a rust finishĪ rust finish can be created by exposing iron or steel to oxygen and moisture. This will cause the metal to react with the sulfur dioxide, nitrogen dioxide, and carbon dioxide in the air to form a thin layer of copper sulfate, copper nitrate, or copper carbonate on the surface of the metal. The lacquer or varnish will also prevent the copper from coming into contact with other metals that could cause it to form a patina.Ī patina finish can be created by exposing copper or bronze to air and moisture. This will create a barrier between the metal and the sulfur dioxide, nitrogen dioxide, and carbon dioxide in the air. Patina can be prevented by coating copper and bronze with a layer of lacquer or varnish. The paint or oil will also prevent the iron from coming into contact with other metals that could cause it to rust. This will create a barrier between the metal and the oxygen and water in the air. Rust can be prevented by coating iron and steel with a layer of paint or oil. How to prevent rustĥ Examples of products with a rust or patina finish Rust is a corrosion product and patina is a protective film.īoth rust and patina can be removed with chemical cleaners, but they will eventually reform if the metal is not protected from the environment. These gases react with the metal to form a thin layer of copper sulfate, copper nitrate, or copper carbonate on the surface of the metal. Patina is caused by the reaction of copper or bronze with sulfur dioxide, nitrogen dioxide, and carbon dioxide in the air. The water molecules break down the oxygen molecules into their component atoms, which then combine with the iron to form rust. Rust is caused by the reaction of iron with oxygen in the presence of water. Patina is a green or blue-green film that forms on copper and bronze when they are exposed to air and moisture. 
Rust is a reddish-brown oxide coating that forms on iron and steel when they are exposed to oxygen and moisture. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
